 
By recognizing that electrons repel each other it is possibleto arrive at geometries which result from valenceelectrons taking up positions as far as possible from each other.The position of the atoms is dictated by the position of thebonding domains of electrons. As indicated in Table I, any compound containing acentral atom with four bonding domains (pairs) of electrons aroundit will exhibit this particular geometry. When four balloons of the same size are tied togetherthe natural arrangement is as a tetrahedron. This was seen in the 'balloon' example we usedin class. The shape of this molecule is a result of the electrons inthe four bonds positioning themselves so as to minimize therepulsive effects. This geometry is a direct result of the repulsionexperienced by the four domains of bonding electrons. The shape wesee is the only possible shape for a central carbon atom withfour bonds. We seethe actual molecular geometry is not flat, but is tetrahedral.This ball and stick model does not adequately represent why themolecule has to have this 3-dimensional arrangement. What does this do to our geometry? Lets rotate this molecule to see what has happened. The Cl-C-Cl bond angles appear to be 90 degrees.However, the actual bond angles in this molecule are 109.5degrees. This is a nice representation of a two dimensional, flatstructure. Here we have a ball andstick model of CCl 4. But this drawing does not tell us about the shape ofthe molecule. Thatis the carbon is the central atom and the four chlorine atoms areterminal. The arrangement of the atoms is correct in my structure. Each chlorineatom has three nonbonding pairs of electrons. In CCl 4 the centralcarbon atom has four bonding domains of electrons. Bonding electrons, which are shared by a pair of atomsand nonbonding electrons, which belong to a particular atom butdo not participate in bonding. Notice that there are two kinds of electron domains in thisstructure. We candraw the Lewis structure on a sheet of paper. Lets consider the Lewis structure for CCl 4. For bent molecular geometry when the electron-pairgeometry is tetrahedral the bond angle is around 105 degrees. For trigonal pyramidal geometrythe bond angle is slightly less than 109.5 degrees, around 107degrees. Note: for bent molecular geometry when the electron-pairgeometry is trigonal planar the bond angle is slightly less than120 degrees, around 118 degrees. The table below summarizes the molecular and electron-pair geometries for different combinations of bonding domains and nonbonding domains of electrons on the central atom. You should note that to determine the shape (molecular geometry) of a molecule you must write the Lewis structure and determine the number of bonding domains of electrons and the number of non-bonding pairs of electrons on the central atom, then use the associated name for that shape. Notice that there are several examples with the same electron-pair geometry, but different molecular geometries. If asked for the electron-pair geometry on the central atom we must respond with the electron-pair geometry. So when asked to describe the shape of a molecule we must respond with a molecular geometry. The molecular geometry is the shape of the molecule. The electron-pair geometry provides a guide to the bond angles of between a terminal-central-terminal atom in a compound. Molecular geometry is the name of the geometry used to describe the shape of a molecule. The term electron-pair geometry is the name of the geometry of the electron-pairs on the central atom, whether they are bonding or non-bonding. In this case there are three domains of electrons around the central atom and the molecualr geometry of the molecule is defined accordingly. 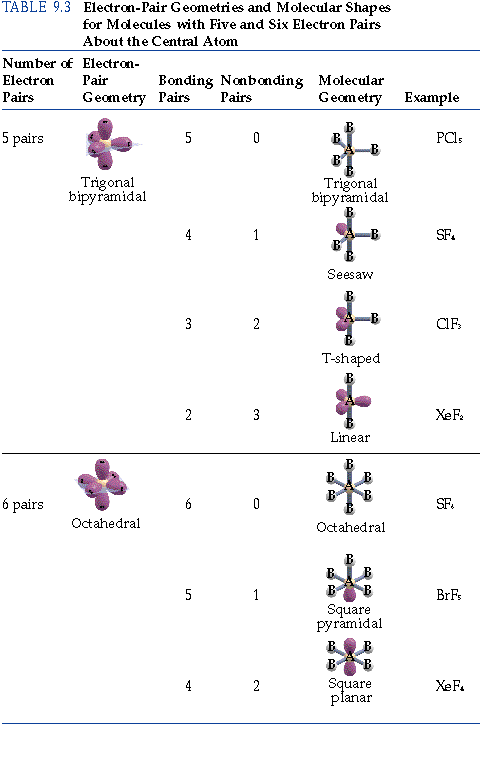
Domain is used when a central atom has two terminal atoms bonded by single bonds and a terminal atom bonded with two pairs of electrons (a double bond). In the table below the term bonding domains (second from the left column) is used in the column for the bonding pair of electrons. Non-bonding pairs of electrons are those pairs of electrons on an individual atom that are not shared with another atom. Bonding pairs of electrons are those electrons shared by the central atom and any atom to which it is bonded. We already have a concept of bonding pair of electrons and non-bonding pairs of electrons. The table below contains several columns. To use the model we will have to memorize a collection of information. This model is fairly powerful in its predictive capacity. We will use a model called the Valence Shell Electron-Pair Repulsion (VSEPR) model that is based on the repulsive behavior of electron-pairs. Molecular Geometry Molecular Geometry VSEPR At this point we are ready to explore the three dimensional structure of simple molecular (covalent) compounds and polyatomic ions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
